The NGIVD Advantage
A cure is only possible
with an accurate diagnosis
One of the biggest bottlenecks facing India’s healthcare system today is the unavailability of fast, accurate, and affordable diagnostics. This is what led NGIVD to develop and pioneer the use of the advanced Multiplexing technology.
Multiplexing technology detects multiple disease biomarkers with just a single sample. The flexibility to use multiple body fluids as samples also allows this novel technology to reach the remotest of location for sample collection and diagnosis. With this technology, it is now possible for people to get an affordable and accurate diagnosis on time thus enabling them to get treatments faster.

Multiplexing & the Science
The Wonder of the Multiplex Technology
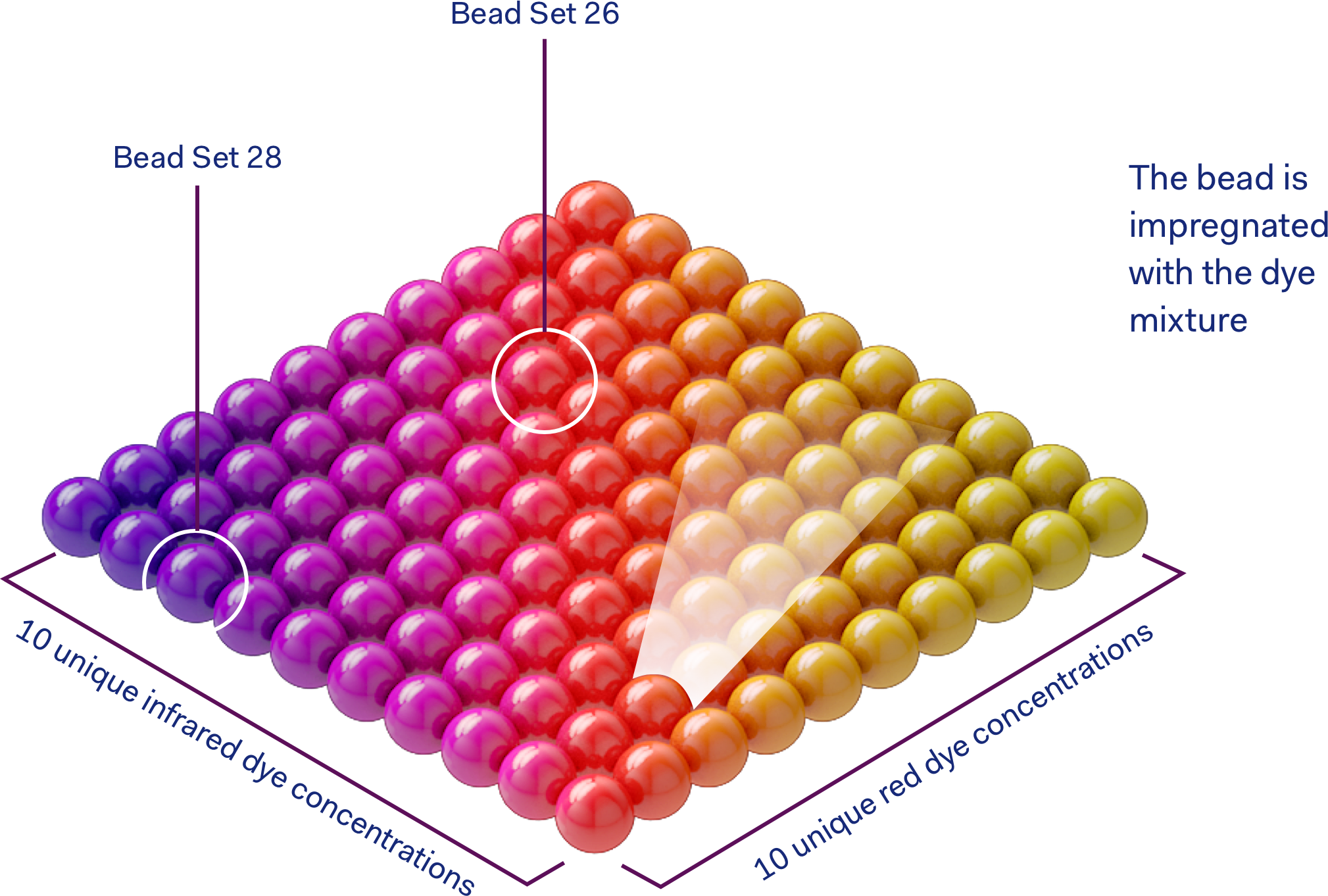
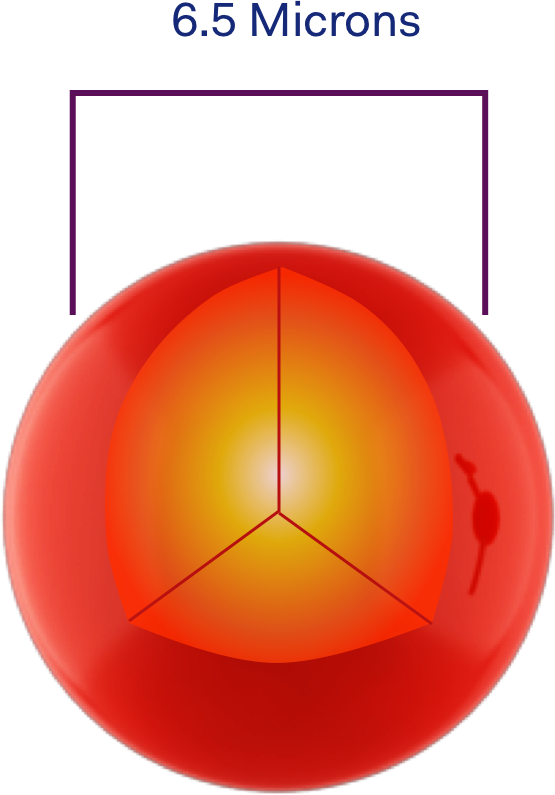
Multiplex Testing uses bead-based conjugation to simultaneously measure multiple pathogens in a single experiment. Multiplexing uses magnetic microsphere or beads of different colours coated with specific antibodies.
Antibodies
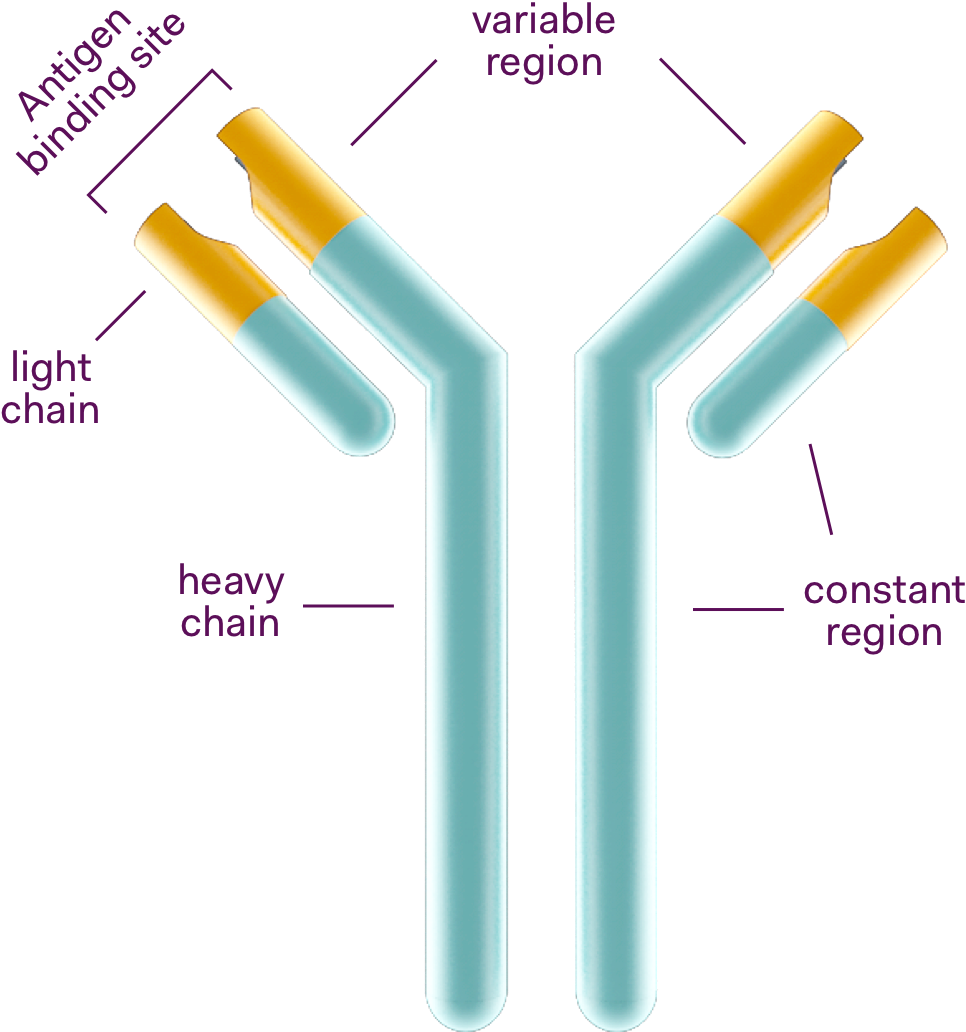
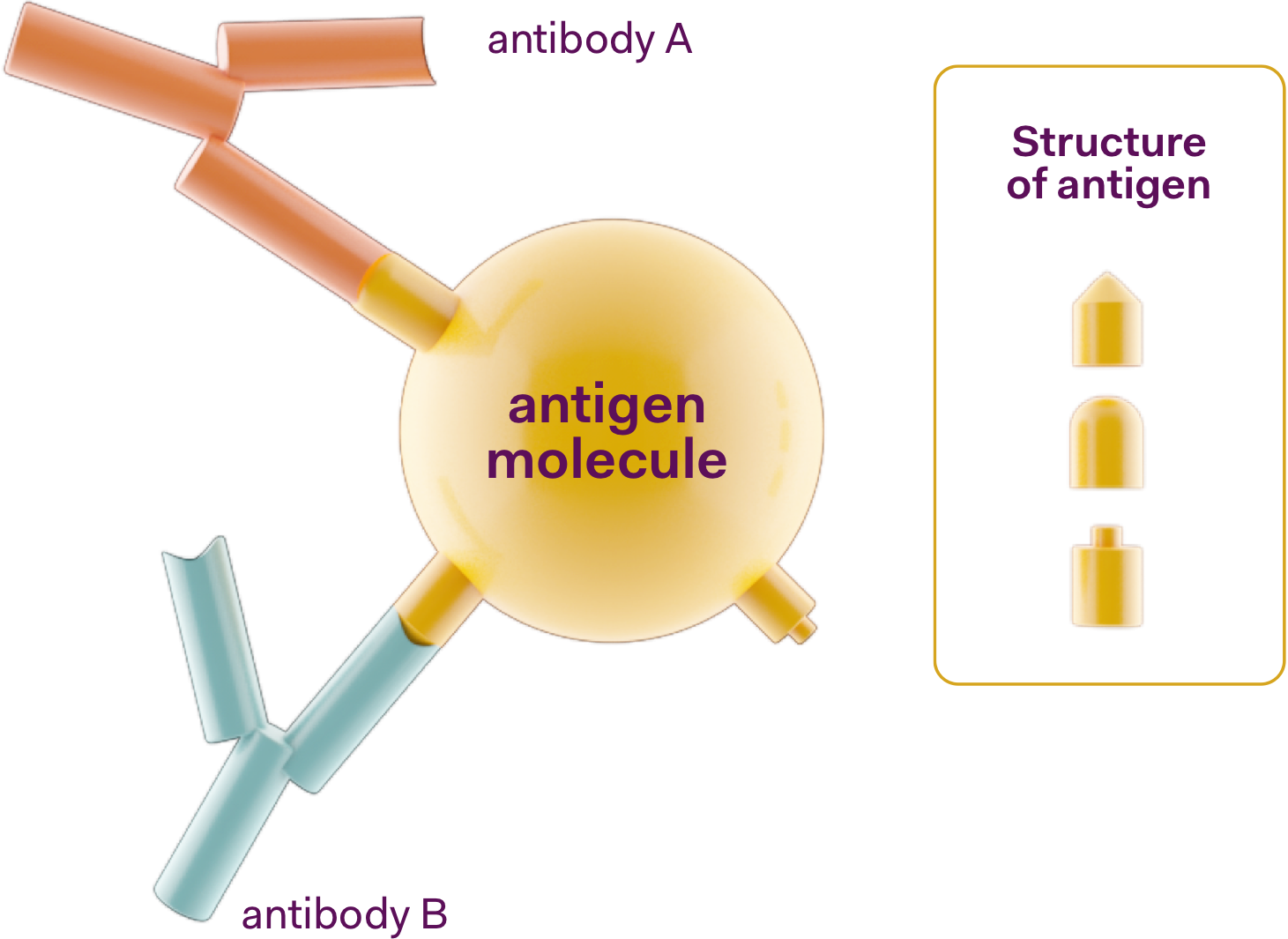
An antibody is a large Y-shaped protein produced by the body to recognise and neutralise a pathogen such as bacteria, virus or microorganism that cause disease. Every unique pathogen will have a corresponding unique antibody. In the body, each antibody binds to a macromolecule of the pathogen and tags it. This allows the body’s immune system to attack the pathogen.
In a Multiplex test, when an antibody binds and tags a macromolecule of the pathogen, it produces a measurable signal. These signals can be detected because they fluoresce under light and the beads are distinguishable by their fluorescent signature.
Advantages of Multiplexing
A Single Sample
Is All It Takes
Using a single sample from the patient, it is now possible with Multiplexing technology to diagnose multiple disease biomarkers in a single test. Not only is the test easy and simple, it is also fast, accurate, and affordable. With easy sample collection techniques like Dried Blood Spot (DBS), samples can be collected even from remote locations through social engagement workers and brought to the nearest lab for analysis.




Science Partners
Pioneering Technology.
Global Recognition.
The science and technology behind our solutions is garnering attention both at home and abroad with grants from BIRAC and accreditation from USISTEF.

BIRAC (Biotechnology Innovation and Research Assistant Council, a Department of Biotechnology) has awarded NGIVD’s TB diagnostic product with a Biotechnology Industry Partnership Program grant.

USISTEF (United States – India Science and Technology Endowment Fund) has also approved NGIVD’s TB diagnostic product.

Licensed the Leishmaniasis LAMP technology from ICMR (Indian Council of Medical Research)